
TONIX PHARMACEUTICALS HOLDING CORP. 8-K
EXHIBIT 99.03

UMass Chan Medical School A Long - Acting Monoclonal Antibody for Seasonal Prevention of Lyme Disease Mark S. Klempner, MD Professor of Medicine March 30, 2026 World Vaccine Congress

UMass Chan Medical School Disclosures Co - Inventor US Patent 10,457,721 Consultant Tonix Pharmaceuticals, Inc Support from N ational C enter for A dvancing T ranslational S ciences (NCATS) NIH, DARPA, DOD Tick Borne Disease Research Program, and NIAID
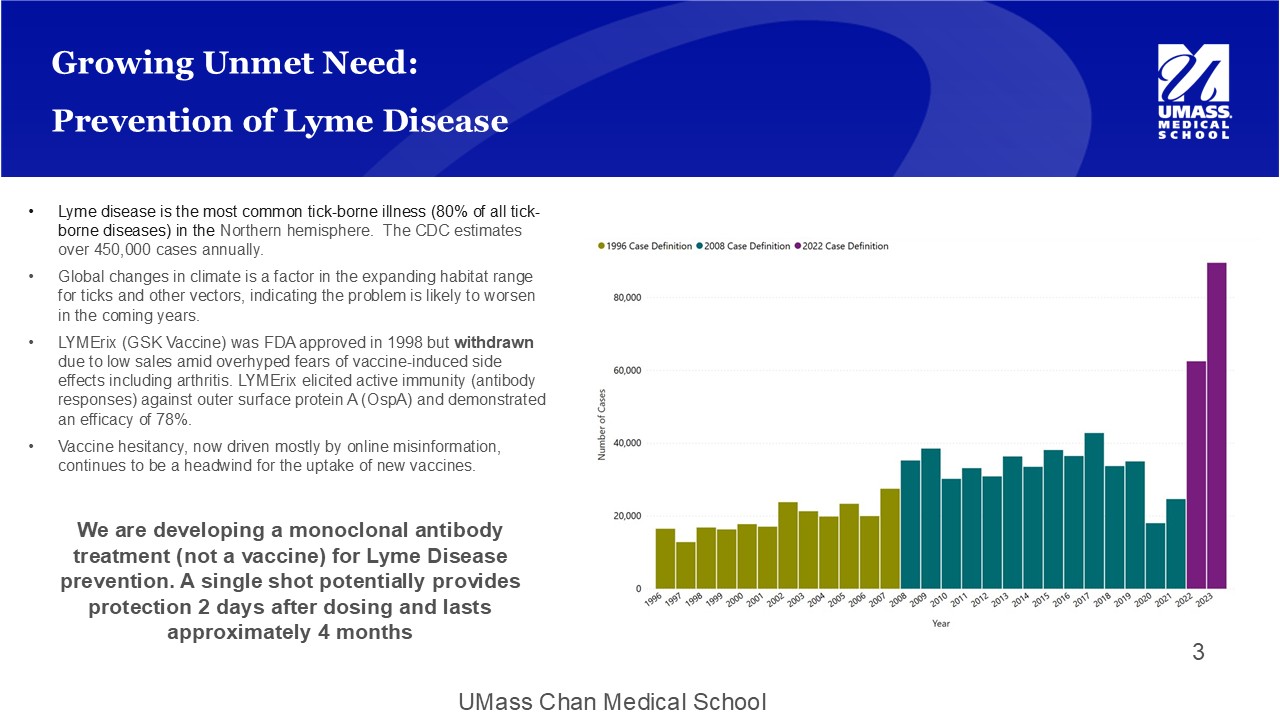
UMass Chan Medical School Growing Unmet Need: Prevention of Lyme Disease • Lyme disease is the most common tick - borne illness (80% of all tick - borne diseases) in the Northern hemisphere. The CDC estimates over 450,000 cases annually. • Global changes in climate is a factor in the expanding habitat range for ticks and other vectors, indicating the problem is likely to worsen in the coming years. • LYMErix (GSK Vaccine) was FDA approved in 1998 but withdrawn due to low sales amid overhyped fears of vaccine - induced side effects including arthritis. LYMErix elicited active immunity (antibody responses) against outer surface protein A ( OspA ) and demonstrated an efficacy of 78%. • Vaccine hesitancy, now driven mostly by online misinformation, continues to be a headwind for the uptake of new vaccines. We are developing a monoclonal antibody treatment (not a vaccine) for Lyme Disease prevention. A single shot potentially provides protection 2 days after dosing and lasts approximately 4 months 3

UMass Chan Medical School 4 Pre - exposure prophylaxis human antibody Person receives 1 injection of a single protective antibody ( red ) at beginning of tick season. Tick carrying the Lyme disease bacteria bites the person and takes in blood containing the protective antibody preventing transmission. Vaccination Person receives 3 injections over 6 months of Lyme bacteria proteins and develops many different antibodies. Tick carrying the Lyme disease bacteria bites the vaccinated person, takes in blood containing multiple antibodies including one (red) that prevents transmission. Lyme bacteria foreign proteins Preventing Lyme Disease Vaccine Compared to Antibody Pre - Exposure Prophylaxis

UMass Chan Medical School 5 Discovery of mAB 221 - 7 with Potent Bactericidal Activity and Coverage of Three OspA Serotypes • Essential for survival while in tick midgut • Anti - OspA immunity is effective in both animals and humans The Target Approach Outer surface protein A ( OspA ) of the Spirochete
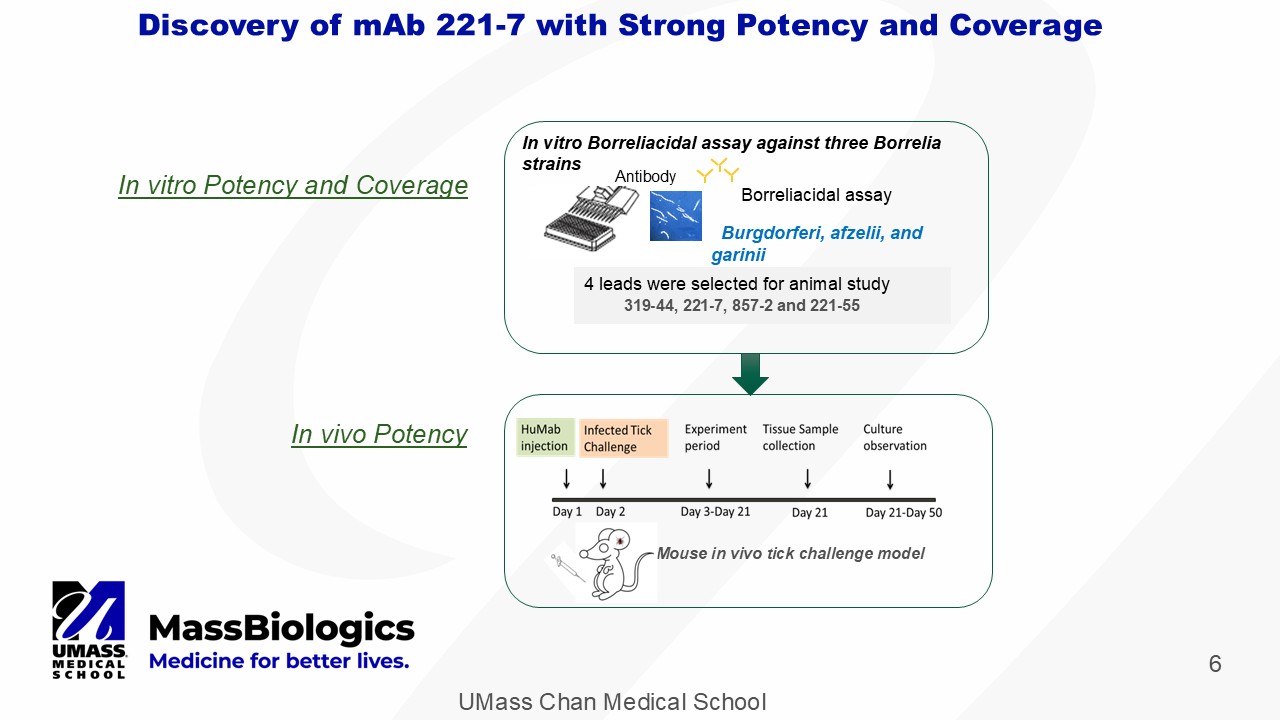
UMass Chan Medical School 6 Discovery of mAb 221 - 7 with Strong Potency and Coverage In vivo Potency In vitro Potency and Coverage Mouse in vivo tick challenge model In vitro Borreliacidal assay against three Borrelia strains Antibody Borreliacidal assay Burgdorferi, afzelii , and garinii 4 leads were selected for animal study 319 - 44, 221 - 7, 857 - 2 and 221 - 55
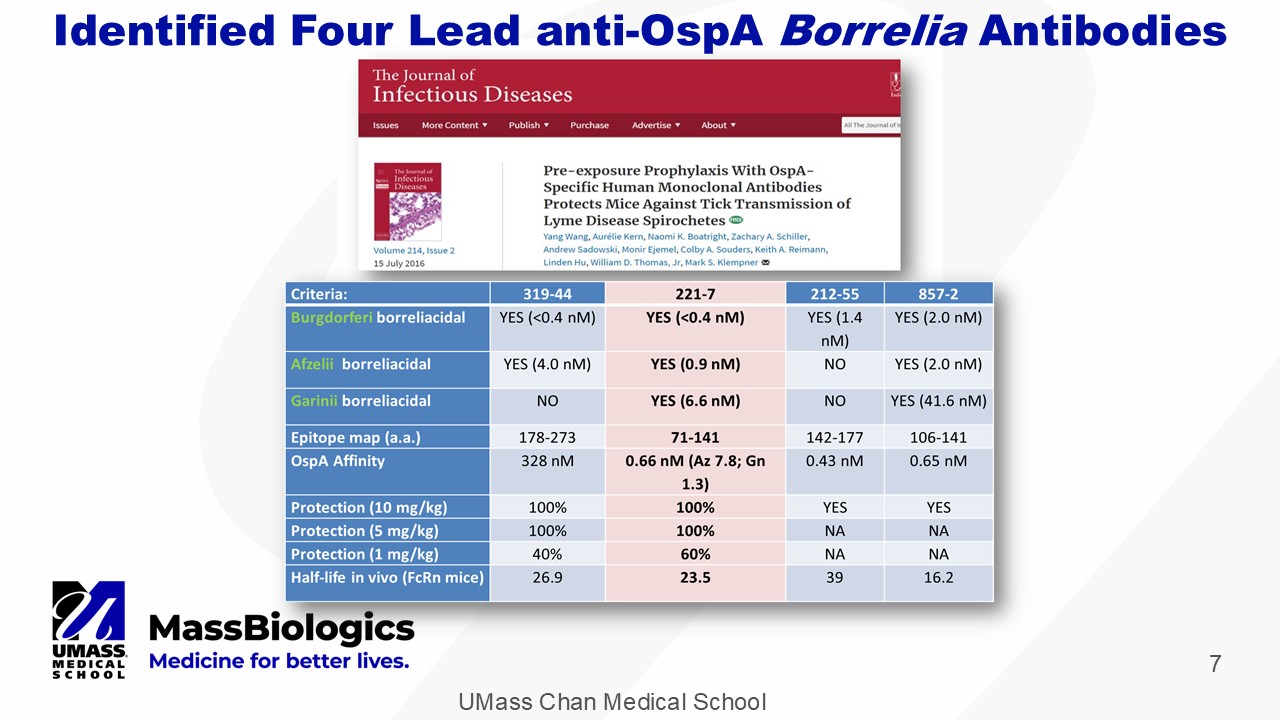
UMass Chan Medical School 7 Identified Four Lead anti - OspA Borrelia Antibodies
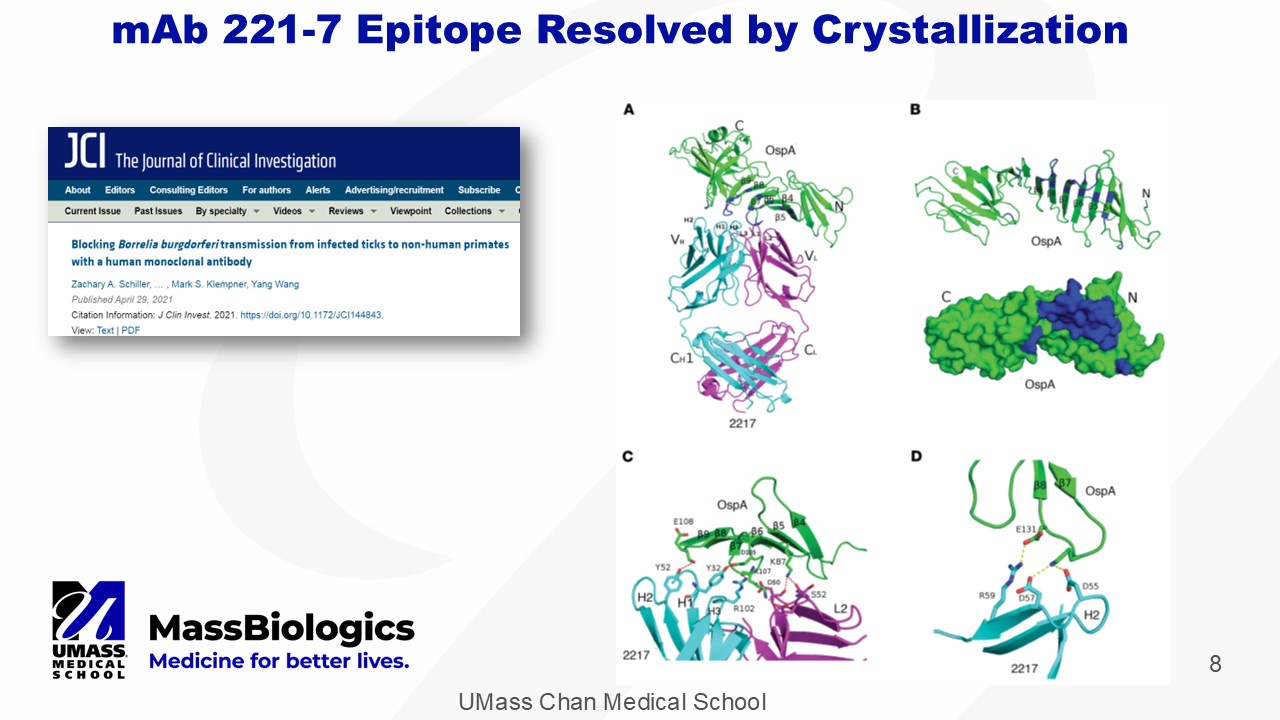
UMass Chan Medical School 8 mAb 221 - 7 Epitope Resolved by Crystallization

UMass Chan Medical School 9 mAb 221 - 7 Does not Recognize the Putative Arthritogenic T - cell Epitope OspA Protein 178 - 273 71 - 141 221 - 7 Epitope 165 - 173 Putative arthritogenic T - cell epitope (YVLEGTLTA), mimicking human leukocyte function - associated antigen - 1 (hLFA - 1) 178 - 273
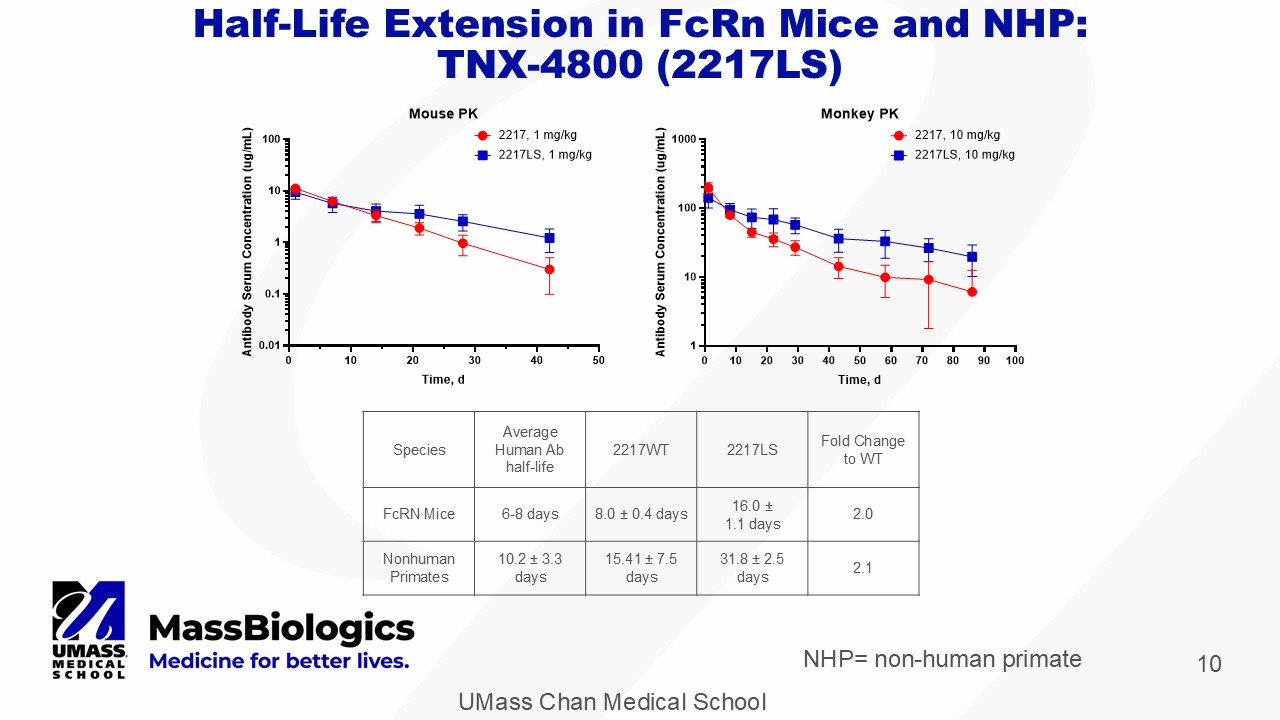
UMass Chan Medical School 10 Fold Change to WT 2217LS 2217WT Average Human Ab half - life Species 2.0 16.0 ± 1.1 days 8.0 ± 0.4 days 6 - 8 days FcRN Mice 2.1 31.8 ± 2.5 days 15.41 ± 7.5 days 10.2 ± 3.3 days Nonhuman Primates Half - Life Extension in FcRn Mice and NHP: TNX - 4800 (2217LS) NHP= non - human primate
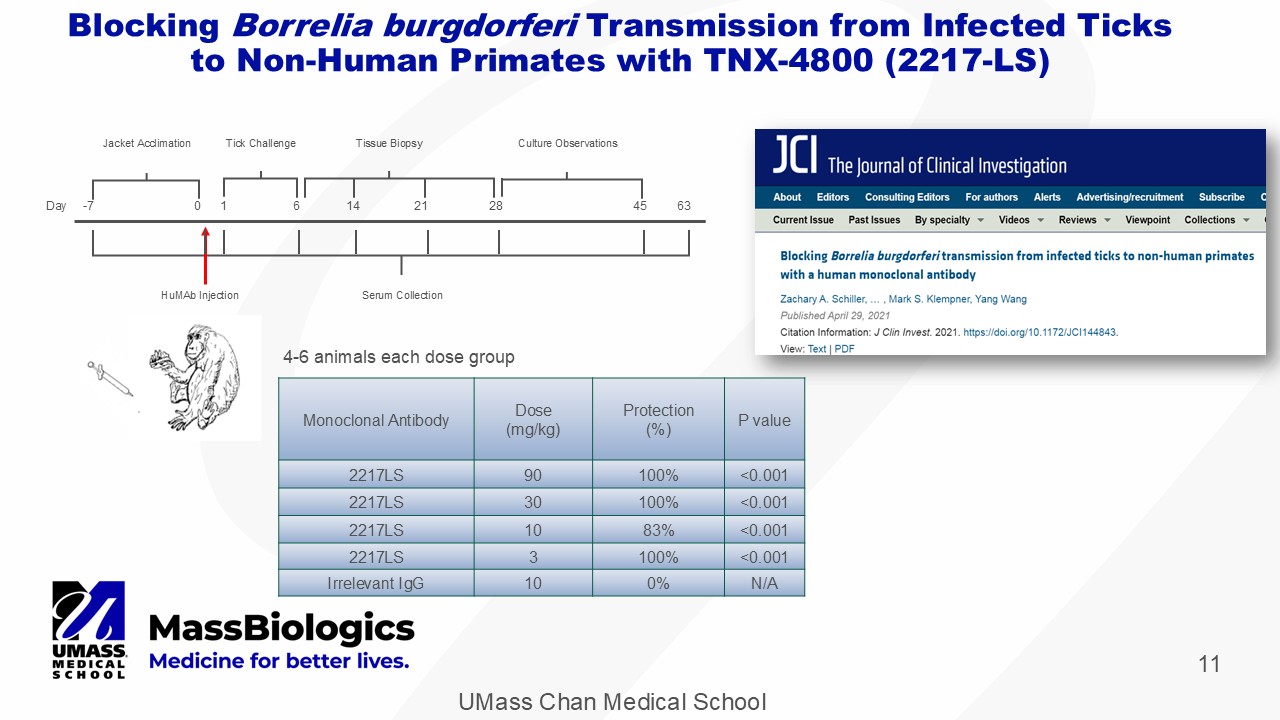
UMass Chan Medical School 11 Blocking Borrelia burgdorferi Transmission from Infected Ticks to Non - Human Primates with TNX - 4800 (2217 - LS) 4 - 6 animals each dose group P value Protection (%) Dose (mg/kg) Monoclonal Antibody <0.001 100% 90 2217LS <0.001 100% 30 2217LS <0.001 83% 10 2217LS <0.001 100% 3 2217LS N/A 0% 10 Irrelevant IgG Day - 7 0 1 6 14 21 28 63 45 Tick Challenge HuMAb Injection Jacket Acclimation Tissue Biopsy Culture Observations Serum Collection
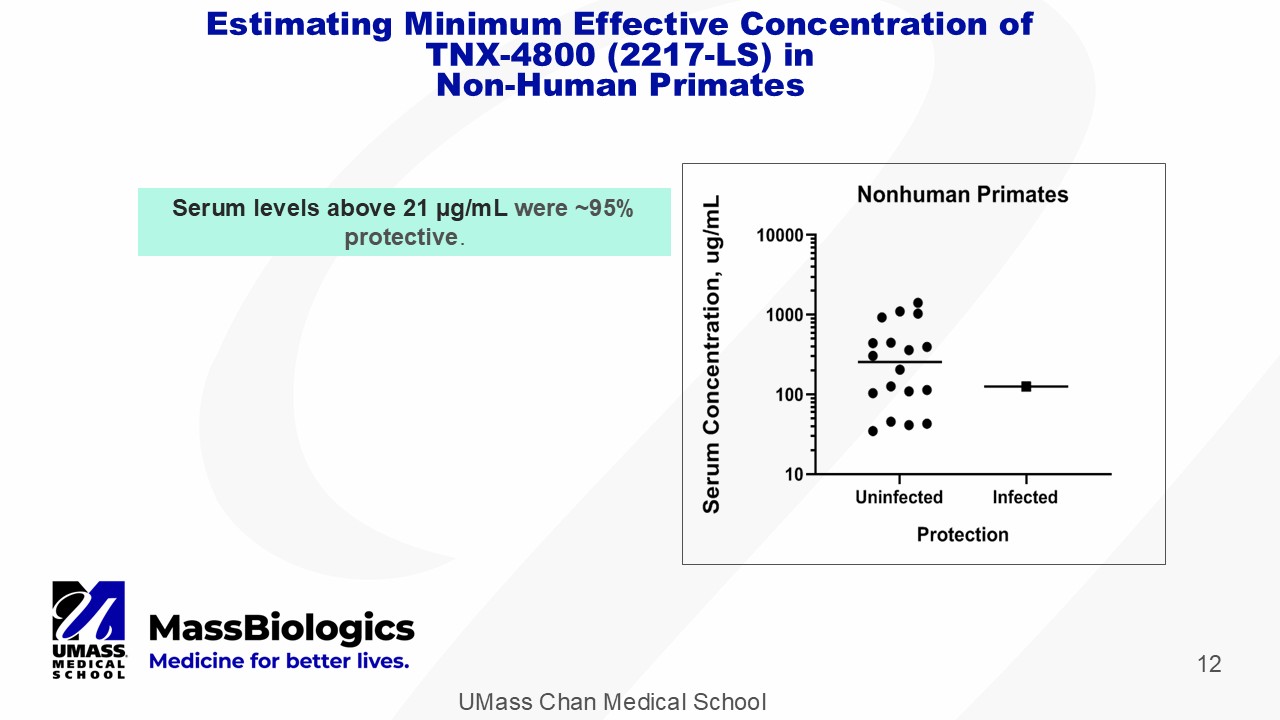
UMass Chan Medical School 12 Serum levels above 21 µg/mL were ~95% protective . Estimating Minimum Effective Concentration of TNX - 4800 (2217 - LS) in Non - Human Primates

UMass Chan Medical School 13 Minimum Effective Concentration (MEC) TNX - 4800 (2217 - LS) • ~ 5 μ g/mL – in vitro bactericidal activity • TNX - 4800 showed EC 50 ≈ 0.56 μ g/mL in vitro 1 • MEC ~ 10X EC 50 • <10 μ g/mL – in vitro tick feeding experiment • TNX - 4800 showed bactericidal activity ≥10 μ g/ml • <21 μ g/mL – in vivo primate challenge models 1,2 • TNX - 4800 serum levels >21 μ g/ml were 95% protective 1 Wang Y, et al. J Infect Dis. 2016 Jul 15;214(2):205 - 11. 2 Schiller ZA, et al. J Clin Invest . 2021;131(11):e144843.

UMass Chan Medical School 14 Nonclinical Safety Data TNX - 4800 ▪ cGLP Tissue Cross Reactivity study in rat and human tissues • No significant cross - reactivity ▪ Non - GLP pharmacokinetic and tick challenge studies in monkeys • Did not reveal a safety signal ▪ cGLP 5 week multiple dose study with 4 week recovery in rats and a cGLP single dose local tolerance study in rats • Observed abnormalities were mild to moderate and all findings were judged non aversive (hematologic and ALT, AST ALP and APTT increases (notably without bilirubin changes), injection site inflammation, liver and spleen organ weight increases, liver histopathology, primarily in males. All findings were reversible). ▪ Exposure in Phase 1 was multiples relative to NOAEL in rats • Starting dose: 63 to 242 – fold lower • Highest dose: 3 to 12 – fold lower NOAEL = No - observed - adverse - effect level

UMass Chan Medical School 15

UMass Chan Medical School Primary Objective : • Evaluate safety and tolerability of a SC injection of TNX - 4800 (2217LS) when administered to healthy subjects Secondary Objective : ▪ Evaluate pharmacokinetics (PK) of a SC dose of TNX - 4800 (2217LS) when administered to healthy subjects Study Population : ▪ Healthy male and female subjects, age 19 to 65 years, inclusive. 44 volunteers enrolled, 41 completed. TNX - 4800 Phase 1 Study Overview Route 2217LS Dose (mg/kg) Treatment N 1 Cohort SC 0.5 2 subjects: placebo 10 subjects: 2217LS 12 1 SC 1.5 2 subjects: placebo 8 subjects: 2217LS 10 2 SC 5 2 subjects: placebo 8 subjects: 2217LS 10 3 SC 10 2 subjects: placebo 8 subjects: 2217LS 10 4 16

UMass Chan Medical School • No significant clinical or laboratory safety signals • The mean exposure, based on AUC - inf and C max for Cohort 4 (10mg/kg), was less than 17% of the highest exposures in the rat toxicology study • Serum TNX - 4800 (2217LS) was measurable at the earliest sampling time of 24 hours indicating rapid absorption. • For all cohorts C max was observed at 10 - 13 days followed by a prolonged elimination phase. • Apparent terminal T ½ after 10 mg/kg dose was 64 days • Max T ½ ranged from 81 - 104 days: ( 10mg/kg - 97 days, 5mg/kg - 87 days, 1.5mg/kg - 104 days, 0.5mg/kg - 81 days) • Cohort 3 (5mg/kg) serum concentrations: • 10 μ g/ml at 4 months (~ in vitro tick - feeding MEC and > in vitro MEC or ~10x EC 50 ) TNX - 4800 Phase 1 Study Results 17
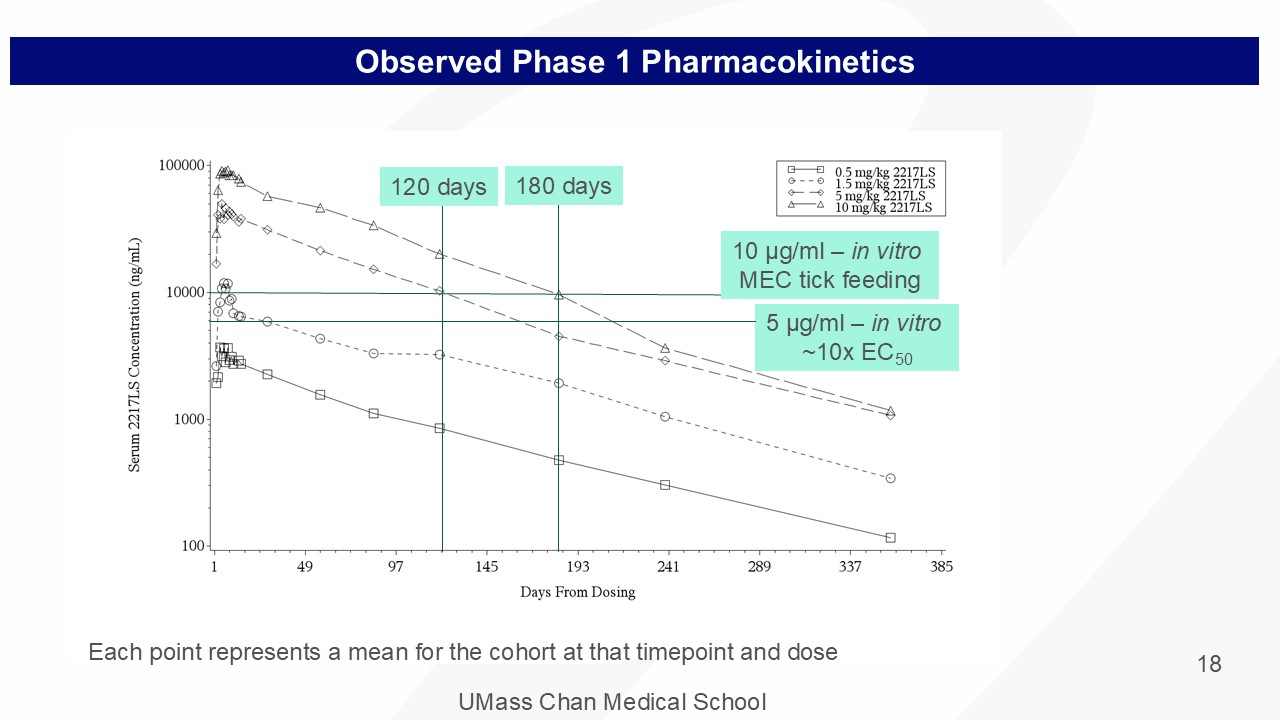
UMass Chan Medical School 18 Observed Phase 1 Pharmacokinetics 5 μ g/ml – in vitro ~10x EC 50 10 μ g/ml – in vitro MEC tick feeding 120 days 180 days Each point represents a mean for the cohort at that timepoint and dose

UMass Chan Medical School • Licensed to Tonix Pharmaceuticals - 2025 now TNX - 4800 • Proposed Phase 2 field study – 2027 pending FDA clearance Next Steps: Phase 2 Field Study 19

UMass Chan Medical School A Long - Acting Monoclonal Antibody for Seasonal Prevention of Lyme Disease Mark S. Klempner, MD Professor of Medicine

UMass Chan Medical School 21 Supplemental Slides
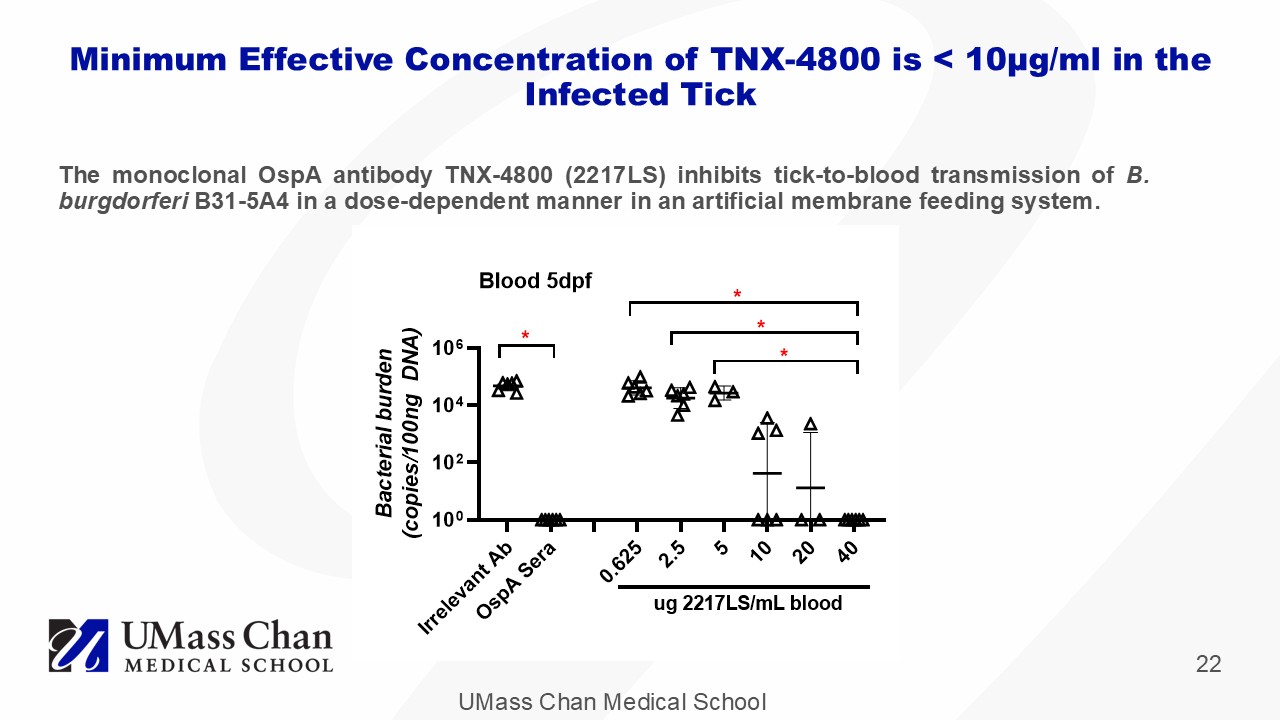
UMass Chan Medical School 22 The monoclonal OspA antibody TNX - 4800 ( 2217 LS) inhibits tick - to - blood transmission of B . burgdorferi B 31 - 5 A 4 in a dose - dependent manner in an artificial membrane feeding system . Minimum Effective Concentration of TNX - 4800 is < 10 μ g/ml in the Infected Tick I r r e l e v a n t A b O s p A S e r a 0 . 6 2 5 2 . 5 5 1 0 2 0 4 0 10 0 10 2 10 4 10 6 B a c t e r i a l b u r d e n ( c o p i e s / 1 0 0 n g D N A ) * * * * ug 2217LS/mL blood Blood 5dpf
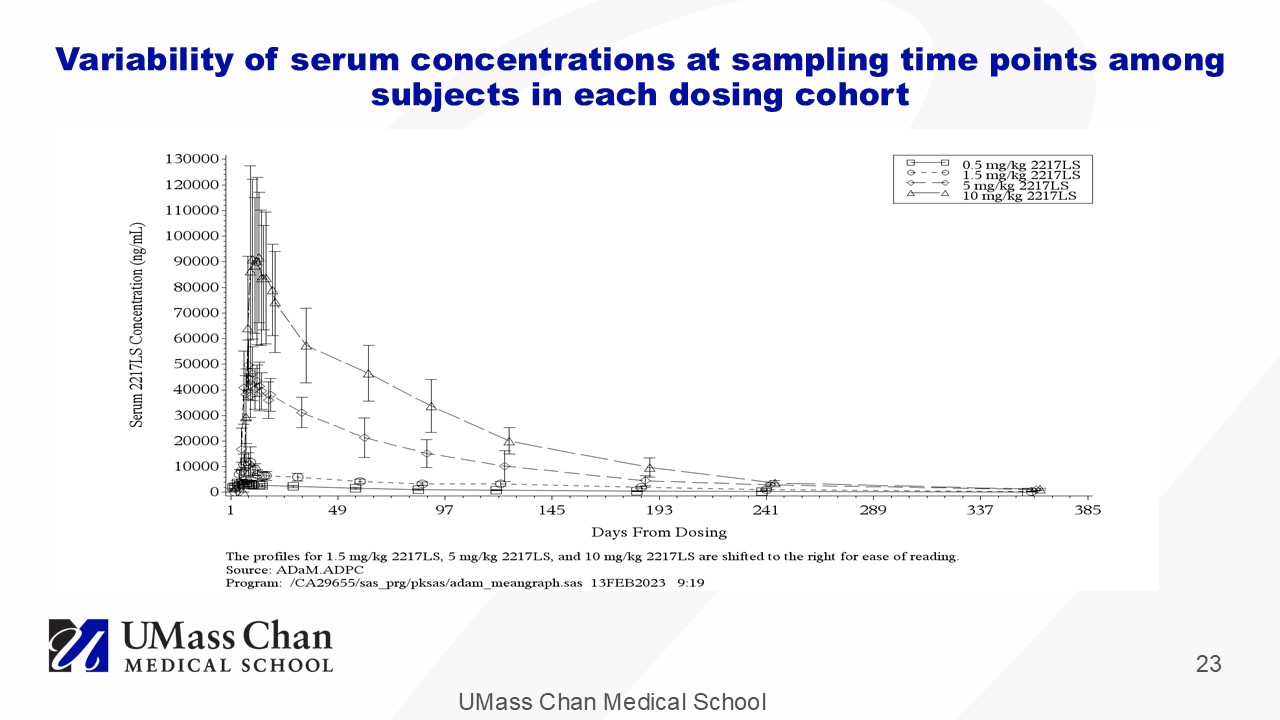
UMass Chan Medical School 23 Variability of serum concentrations at sampling time points among subjects in each dosing cohort
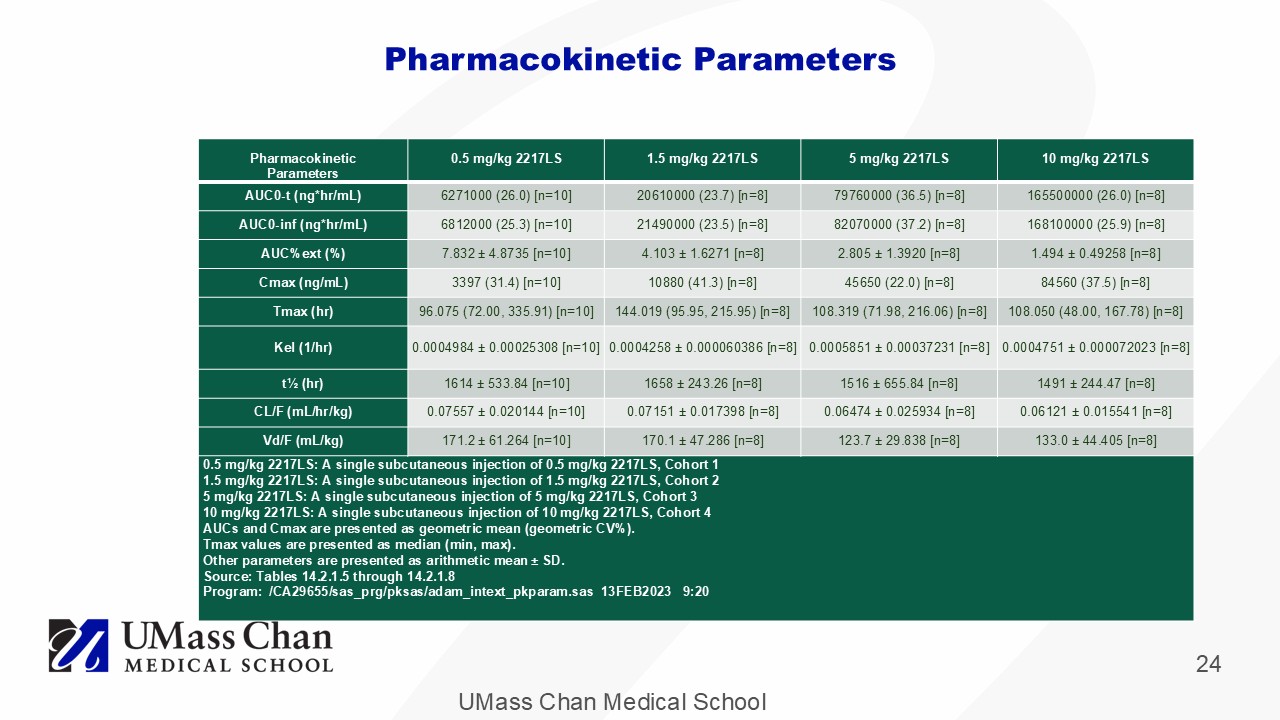
UMass Chan Medical School 24 Pharmacokinetic Parameters 10 mg/kg 2217LS 5 mg/kg 2217LS 1.5 mg/kg 2217LS 0.5 mg/kg 2217LS Pharmacokinetic Parameters 165500000 (26.0) [n=8] 79760000 (36.5) [n=8] 20610000 (23.7) [n=8] 6271000 (26.0) [n=10] AUC0 - t (ng* hr /mL) 168100000 (25.9) [n=8] 82070000 (37.2) [n=8] 21490000 (23.5) [n=8] 6812000 (25.3) [n=10] AUC0 - inf (ng* hr /mL) 1.494 ± 0.49258 [n=8] 2.805 ± 1.3920 [n=8] 4.103 ± 1.6271 [n=8] 7.832 ± 4.8735 [n=10] AUC%ext (%) 84560 (37.5) [n=8] 45650 (22.0) [n=8] 10880 (41.3) [n=8] 3397 (31.4) [n=10] Cmax (ng/mL) 108.050 (48.00, 167.78) [n=8] 108.319 (71.98, 216.06) [n=8] 144.019 (95.95, 215.95) [n=8] 96.075 (72.00, 335.91) [n=10] Tmax ( hr ) 0.0004751 ± 0.000072023 [n=8] 0.0005851 ± 0.00037231 [n=8] 0.0004258 ± 0.000060386 [n=8] 0.0004984 ± 0.00025308 [n=10] Kel (1/hr) 1491 ± 244.47 [n=8] 1516 ± 655.84 [n=8] 1658 ± 243.26 [n=8] 1614 ± 533.84 [n=10] t½ ( hr ) 0.06121 ± 0.015541 [n=8] 0.06474 ± 0.025934 [n=8] 0.07151 ± 0.017398 [n=8] 0.07557 ± 0.020144 [n=10] CL/F (mL/ hr /kg) 133.0 ± 44.405 [n=8] 123.7 ± 29.838 [n=8] 170.1 ± 47.286 [n=8] 171.2 ± 61.264 [n=10] Vd /F (mL/kg) 0.5 mg/kg 2217LS: A single subcutaneous injection of 0.5 mg/kg 2217LS, Cohort 1 1.5 mg/kg 2217LS: A single subcutaneous injection of 1.5 mg/kg 2217LS, Cohort 2 5 mg/kg 2217LS: A single subcutaneous injection of 5 mg/kg 2217LS, Cohort 3 10 mg/kg 2217LS: A single subcutaneous injection of 10 mg/kg 2217LS, Cohort 4 AUCs and Cmax are presented as geometric mean (geometric CV%). Tmax values are presented as median (min, max). Other parameters are presented as arithmetic mean ± SD. Source: Tables 14.2.1.5 through 14.2.1.8 Program: /CA29655/ sas_prg / pksas / adam_intext_pkparam.sas 13FEB2023 9:20

UMass Chan Medical School 25 Outcome • Two - thirds of parents were willing to vaccinate their child against LD (68.0% definitely/probably would, 18.4% unsure, 13.7% definitely/probably would not) • Parental willingness to be vaccinated against LD was highly correlated with willingness for their child in. • Vaccine safety concerns were among the top reasons for LD vaccine hesitancy • More parents preferred pre - formed antibody (42.4%) over 3 - dose vaccine series (36.2%) • Significant predictors of preference for monoclonal antibody included prior awareness of LD, living in a rural area, and positive attitude towards vaccines CDC Study Makes a Strong case for Antibody vs Vaccine Methods • Conducted online survey (N=1,351) of parents of children aged 5 – 18 years in states with high or emerging Lyme Disease (LD) incidence. • Primary outcome was willingness (definitely / probably would vs unsure or definitely / probably would not) for their child to receive an LD vaccine. • Secondary outcome was preference for annual monoclonal antibody injections vs. a 3 - dose vaccine series with boosters every few years. Lyme Disease Vaccine Intentions Among Parents of 5 – 18 Year Olds in U.S. States with High or Emerging Incidence (CDC - sponsored study presented at 2023 Pediatric Academic Societies (PAS) Conference) CDC = U.S. Centers for Disease Control